Crack act organic compounds. EPA Moves To Crack Down On Toxic Emissions From Oil Refineries 2019-05-06
EPA Moves To Crack Down On Toxic Emissions From Oil Refineries

Once the compounds are sorted, students will be asked to make and record an observation that can be used to identify an organic compound. Nylon used to make B rope, C umbrellas, and D rain jackets. . They also need to be able to evaluate nutritional supplements because providing the wrong one can get an athlete banned from competition and may bring sanctions against a school. The hydrogen bond in the alcohol means it can soluble into the water. We can calculate a more general bond energy by finding the average of the bond energies of a specific bond in different molecules to get the average bond energy.
12: Organic Chemistry

It can be used to determine which Lewis Dot Structure is most suitable when there are multiple Lewis Dot Structures. In Wikipedia, The Free Encyclopedia. The alcohol contain five and more carbon are insoluble in water as the carbon chain has became the dominance in the molecule. In the following activity, you will check your understanding by identifying examples of organic compounds. In cases like this, start trying to balance the equation, by changing the cofactor in front of the alkane to 2. Like other science-based professions, their work requires knowledge, ingenuity, and complex thinking, as well as a great deal of technical skill. Today organic chemistry has been reclassified as the study of compounds that contain carbon, and inorganic chemistry is the study of the chemistry of all other elements.
CH105: Chapter 7

There are 6 hydrogens that could get replaced on the end carbon atoms compared with only 2 in the middle. Thus, cracking can be employed to improve the yield of gasoline. In the substitution reaction, a hydrogen atom in the methane is replaced by a chlorine atom. These are high-energy bonds that store energy from the metabolism of foods. Both hydrogen bonding networks extend in all directions. When these two compounds polymerize, the amine and acid groups combine, each time with the loss of a molecule of water. For example, with alkanes, the ones with an even number of carbon atoms are marginally harder than those with an odd number! The cubes were set in motion from a fixed starting angle, and the period for each was recorded.
Deterioration of cement

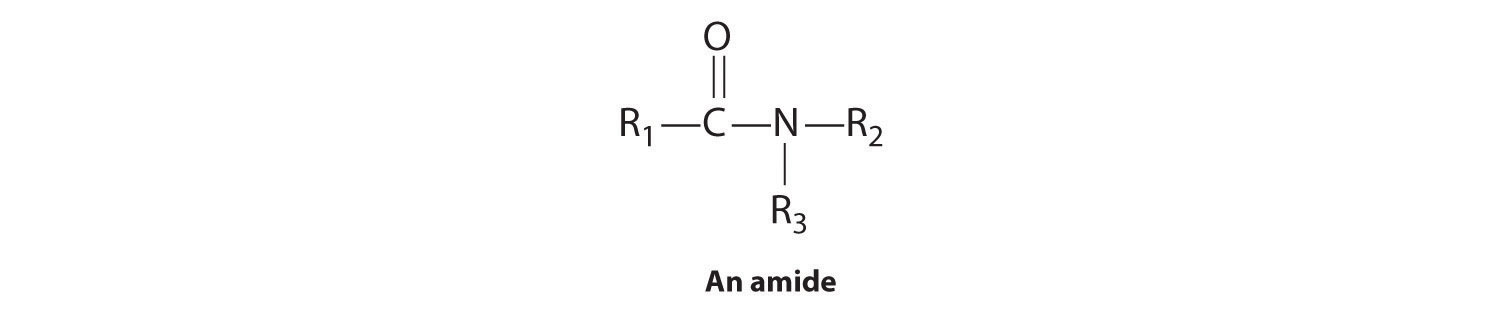
This leaves the user in a pleasureless state and often craving more cocaine. If you said 19, you are correct again. The recommended courses for preparation in this field include anatomy, physiology, medical terminology, and—not surprisingly—chemistry. The formation of esters and the related compounds, thioesters, amides, and phosphoesters are formed by dehydration synthesis, involving the loss of water. If one or both of the two remaining bonds on the atom are attached to alkyl or aryl groups, the compound is a substituted amide. For example, if only 4 moles of oxygen was present, the combustion of one mole of propane would produce only 1 mole of carbon dioxide and 2 moles of carbon monoxide. The other 5% may include a limited number of non-organic materials that are essential to organic processing and not available in an organic form.
SAT Chemistry Subject Test: Practice tests and explanations_www.littleboyblu.com

This presents a much more serious fire hazard than a natural-gas leak because it is more difficult to rid the room of the heavier gas. Equipment for such shutdown operations must be installed at the gantry. Like most volatile amines, aniline possesses the odor of rotten fish. If you use bromine instead of chlorine, the great majority of the product is where the bromine is attached to the center carbon atom. Athletic trainers work in high schools, colleges, and other organizations where athletics programs are found. Thiols resemble the alcohol functional group and can undergo oxidation with other thiols to form disulfide bonds. Which two compounds are not isomers of each other? The external wall and roofs of tanks above ground must be painted in a colour with a total radiant heat reflectance of 70% or more.
51.7 CRACKING AND REFORMING ORGANIC COMPOUNDS

First, students answer several multiple choice questions identifying organic compounds and their components. Alkanes are generally unreactive toward laboratory acids, bases, oxidizing agents, and reducing agents. These forces to do not release a sufficient amount of energy to compensate for the energy required to break the hydrogen bonds in water. It has to do with the stability of the carbon radical intermediate that forms during the reaction. People who swallow gasoline or other liquid alkane mixtures should not be made to vomit, as this would increase the chance of getting alkanes into the lungs.
SAT Chemistry Practice Test: Organic Chemistry_www.littleboyblu.com

Consider butane as an example: Neither positive ions nor negative ions are attracted to a nonpolar molecule. Which compound below belongs to the same class of hydrocarbons? Provided the combustion is complete, all the hydrocarbons will burn with a blue flame. Other matters may be taken into account by the Agency 29. However, sulfur is not as electronegative as oxygen, thus, intermolecular forces between sulfur-containing molecules is similar but weaker than those containing oxygen. Carbon monoxide can also bind to hemoglobin.
Local Farmers Using Certified Organic Farming Practices To Ensure Products Are Chemical Free

In the case of the alkanes, these are the London dispersion forces. Many heterocyclic amines occur naturally in plants. You are convinced you have discovered a new organic compound and are explaining your discovery to another scientist. Allow students to use their Periodic Table as a reference tool. The process involves the cleavage of carbon-carbon and carbon-hydrogen bonds and results in the formation of molecules of lower hydrocarbons. These compounds have both polar and nonpolar groups, enabling them to bridge the gap between water-soluble and water-insoluble phases.
SAT Chemistry Practice Test: Organic Chemistry_www.littleboyblu.com
For an exothermic chemical reaction, energy is given off as reactants are converted to products. Examples include gasoline, plastics, detergents, dyes, food additives, natural gas, and medicines. In this chapter, we will focus on compounds that contain nitrogen, suflur, and phosphorous. Remember that energy can be evaluated in kcal or kJ and that the conversion factor between the two is: 4. Their critical duties often include rescue work and emergency medical procedures in a wide variety of settings, sometimes under extremely harsh and difficult conditions. The sulfur mustard in the opening essay is an example of a thioether. Depiction of disulfide protein bridges in C, provided by: Disulfide Bonds in Hair and Feathers — A Closer Look Over 90% of the dry weight of hair comprises proteins called keratins, which have a high disulfide content, from the amino acid cysteine.