Define helmholtz free energy. What does Helmholtz free energy mean? 2019-03-14
Helmholtz
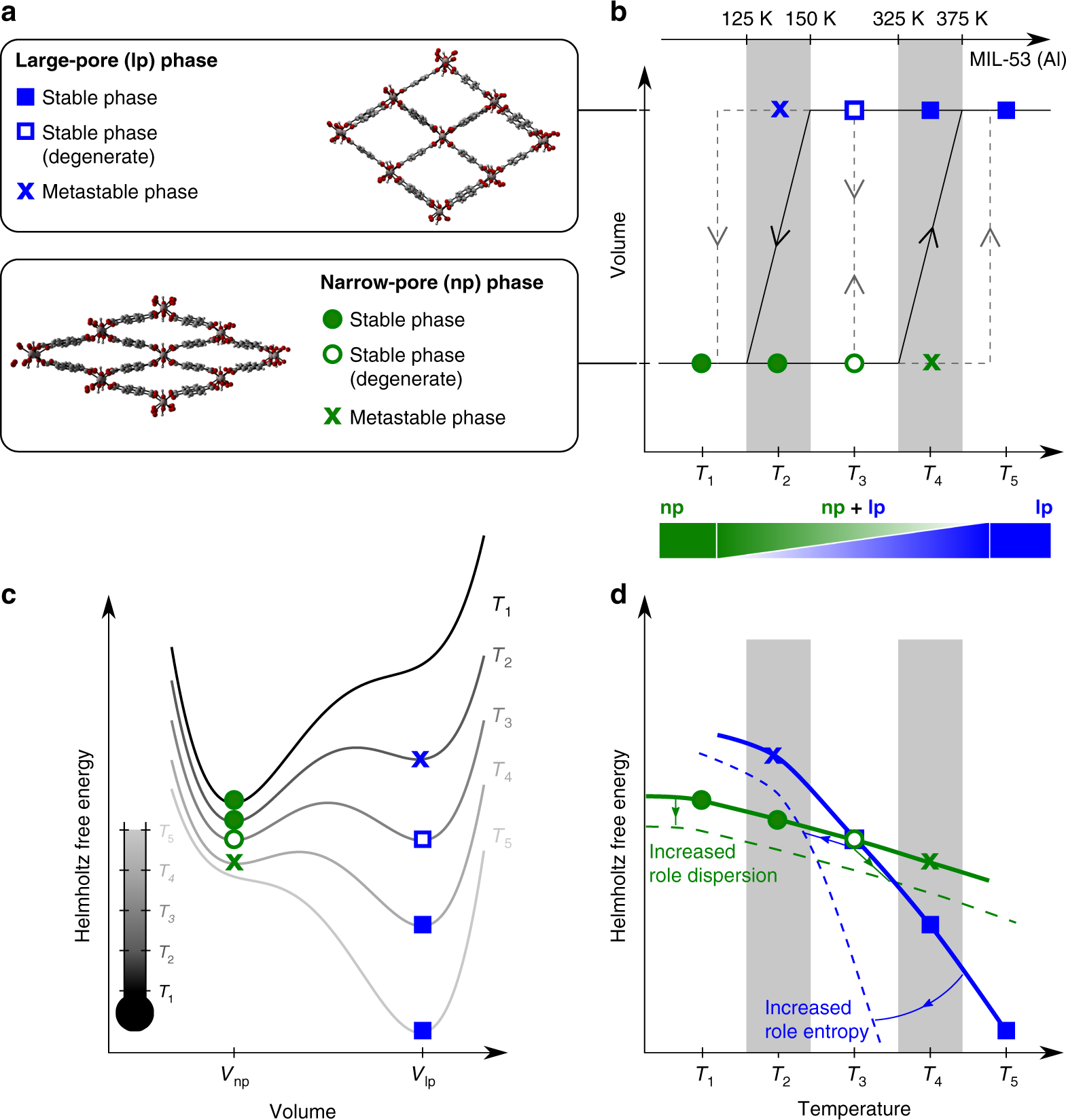
For an Isothermal-isobaric transformation, G is constant. In case there are other external parameters the above equation generalizes to: Here the are the external variables and the the corresponding. We said after the introduction of the first law which introduced the internal energy, U that we would be introducing three more functions that have units of energy. If there's a mathsy possible understanding that might be preferred, as I have more of a maths background. Thus Since A is a state function, this equation is true for any path connecting the same initial state V,T and final state V+dV,T+dT.
Helmholtz free energy definition
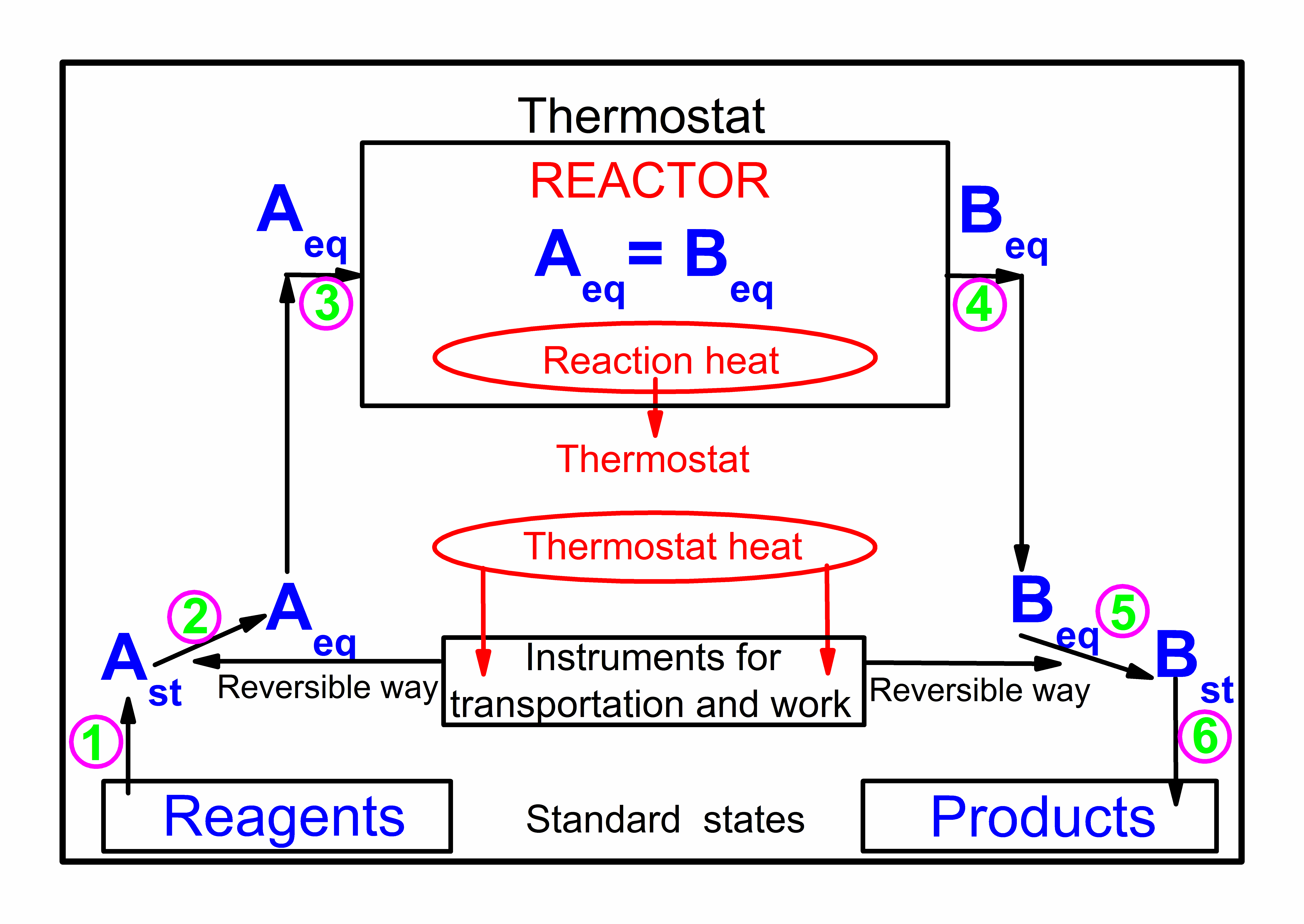
We are interested in processes which provide useful energy in the form work to the surroundings, i. No, because in practice, systems have different constraints imposed on them, and to understand them we need to find the physical principles that describe their behavior. We denote the canonical probability distributions for the Hamiltonian and the trial Hamiltonian by and , respectively. The Gibbs' free energy is a thermodynamic function of the natural variables, temperature T and pressure P. In physics, the letter F can also be used to denote the Helmholtz energy, as Helmholtz energy is sometimes referred to as the Helmholtz function, Helmholtz free energy, or simply free energy.
Gibbs Helmholtz equation in chemistry by
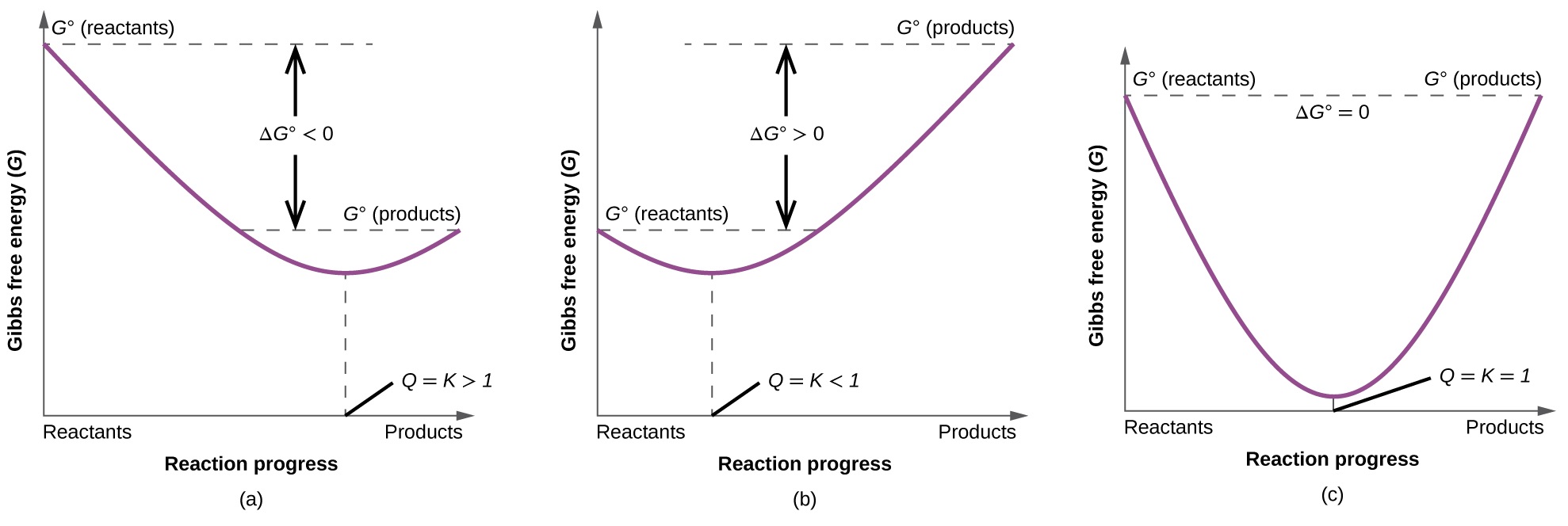
The Bogoliubov inequality is often formulated in a sightly different but equivalent way. In the case of linear materials that obey , the stress is related to the strain by: where we are now using for the tensors, in which repeated indices in a product are summed. . An example is , the amount of increase of free energy when the area of surface increases by every unit area. We now know that these functions are H, A, and G. If we choose this trial Hamiltonian such that where both averages are taken with respect to the canonical distribution defined by the trial Hamiltonian , then where is the free energy of the original Hamiltonian and is the free energy of the trial Hamiltonian.
Helmholtz and Gibbs Free Energies

If we now substitute in here the expressions for the probability distributions: and we get: Since the averages of and are, by assumption, identical we have: Here we have used that the partition functions are constants with respect to taking averages and that the free energy is proportional to minus the logarithm of the partition function. The Gibbs free energy G is defined by The internal energy U might be thought of as the energy required to create a system in the absence of changes in temperature or volume. Hence its utility to ; and to -phase chemists and engineers, who do not want to ignore p dV work. In that case, you don't have to put in as much energy. Recall that we can control temperature, pressure, and volume. Because the person changed the stationary position of the box, that person exerted energy on that box.
a
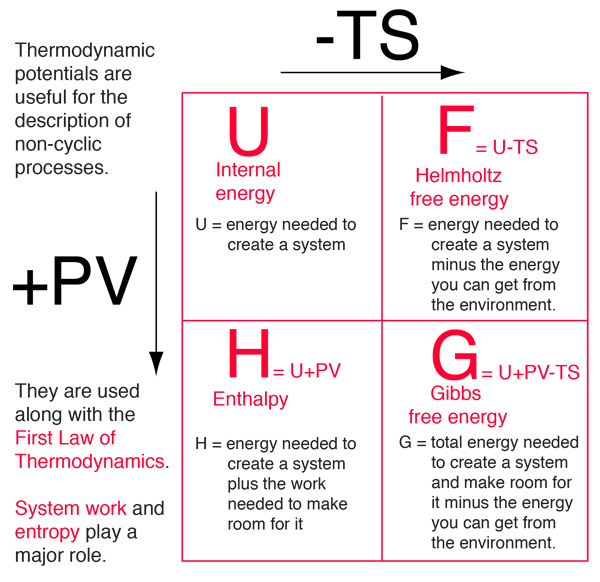
It stays constant during any isothermal-isochoric change. Gibbs free energy is the maximum reversible work that can be obtained from a system. In case of a spontaneous change at constant T and V, the last term will thus be negative. We denote the diagonal components of the density matrices for the canonical distributions for and in this basis as: and where the are the eigenvalues of We assume again that the averages of H and in the canonical ensemble defined by are the same: where The inequality still holds as both the and the sum to 1. Gibbs free energy is a relative measurement of the stability of a phase at Constant Temperature and Pressure. The different free energies are Legendre transformations with respect to your internal energy variables of state, and they correspond to cases where the system can now share that variable with some environment which is so big that it maintains a constant partial derivative with respect to that variable. The quintessential concept in the application of the laws of thermodynamics is how to derive that useful quantity from the second law.
Helmholtz free energy : definition of Helmholtz free energy and synonyms of Helmholtz free energy (English)
Several free energy functions may be formulated based on system criteria. By 1865, the German physicist had shown that this equivalence principle needed amendment. Also, I am unclear as to why these two potentials are so useful and prevalent. In 1847, the English physicist showed that he could raise the temperature of water by turning a paddle wheel in it, thus showing that heat and mechanical work were equivalent or proportional to each other, i. This is equal to the difference between the initial energy and the final energy.
What are Helmholtz free energy and Gibbs free energy?
Translated from Russian by J. The Gibbs Free Energy and Helmholtz Free Energy Next: Up: Previous: The entropy gives a maximal principle, but for the entire universe. Chemical Reactions Gibbs Free Energy:Chemical reactions are spontaneous when the Gibbs free energy change is negative. This equation is then again valid for both reversible and non-reversible changes. While Gibbs free energy is most commonly used as a measure of thermodynamic potential, especially in the field of chemistry, the isobaric restriction on that quantity is inconvenient for some applications.
What are Helmholtz free energy and Gibbs free energy?

The probability to find the system in some energy eigenstate r is given by: where Z is called the of the system. If we wish to describe phenomena like chemical reactions, then the best we can do is to consider suitably chosen initial and final states in which the system is in metastable thermal equilibrium. Changes in the Gibbs free energy G correspond to changes in free energy for processes at constant temperature and pressure. Then, the above equation can be modified as below. Free energy is subject to loss in the course of such work. From the , for a we may say that. T and V are much more convenient variables than S and V.
The Gibbs Free Energy and Helmholtz Free Energy

In 1873, published A Method of Geometrical Representation of the Thermodynamic Properties of Substances by Means of Surfaces, in which he introduced the preliminary outline of the principles of his new equation able to predict or estimate the tendencies of various natural processes to ensue when bodies or systems are brought into contact. In solution chemistry, on the other hand, most chemical reactions are kept at constant pressure. You can complete the definition of Helmholtz free energy given by the English Definition dictionary with other English dictionaries: Wikipedia, Lexilogos, Oxford, Cambridge, Chambers Harrap, Wordreference, Collins Lexibase dictionaries, Merriam Webster. After the spontaneous change, the system, as described by thermodynamics, is a different system with a different free energy function than it was before the spontaneous change. For example, one doesn't usually state that ice will freeze at a particular temperature and pressure because the entropy of the ice plus the freezer plus the wires that run to the power station plus the power station, etc, have a total sum entropy that increases. Helmholtz free energy and Gibbs free energy according to Wikipedia However, according to Wikipedia, Helmholtz free energy is a thermodynamic potential that measures the useful work obtainable from a closed thermodynamic system at a constant temperature and volume isothermal, isochoric. The above relation may be derived from the internal energy function by means of Legendre's transformations to change variables.