History of phone batteries. History and Timeline of the Battery 2019-04-12
Battery History

They're really smaller but easy to press and I appreciated a relaxed typing knowledge in my transient hands-on -- but I am going to observe how it genuinely handles inside the total evaluation. Cockrell School of Engineering, The University of Texas at Austin One of the co-inventors of the original lithium-ion battery himself is taking another tack toward solid-state electrolytes: John Goodenough, emeritus professor of engineering at the University of Texas, and filed a for a battery with a glass-based electrolyte. Chiang and his team were faced with the necessity to quickly decide whether to continue focusing on the original self-assembly concept or to propel A123 into the commercial world more quickly by incorporating the new electrode material into conventional mechanically assembled lithium-ion batteries. Based on digital technology, it focused on interoperability across national boundaries and consequent different frequency bands, call quality and low costs. Ten hours of recharge would result in approximately 30 minutes to an hour of talk time. But silicon tends to expand as it absorbs lithium, so batteries will have to account for that.
History of the battery

We do not guarantee individual replies due to extremely high volume of correspondence. But why is battery technology lagging and why doesn't it seem to be getting any better? Materials in this case are very small particles. This simpler version dispensed with the porous barrier. A , the first chemical battery provided the main source of before the development of and around the end of the 19th century. In the rechargeable batteries inside today's smartphones, lithium is found in the electrolyte, but not in the anode, restricting the efficiency and lifespan of the blocks. The most obvious example of this is the Apple iPhone and subsequent competitors like the Droid models.
History of the battery

It was Alessandro Volta who invented the first true battery in 1800. An example of a reaction that produces electrons is the oxidation of iron to produce rust. Sweden, Norway, Finland, and Denmark also received 1G that same year. G of Android phones, it was a long way from the high-end Android smartphones we use today. The reaction caused the lead anode to release electrons, while the reaction at the lead dioxide end consumed the electrons, causing a flow of electric current.
Battery History

The Voltaic Pile produced a continuous and stable current and did not lose much charge over time or when not being used. The phone could last for about 30 minutes of talk time. Mobile devices were designed to be powered by lightweight energy storage systems. Using powdered zinc gave the anode a greater surface area. It is preferred to use iron as the positive electrode, and as exciting-liquid nitro muriatic acid, aqua regis, the mixture consisting of muriatic and nitric acids.
Battery History
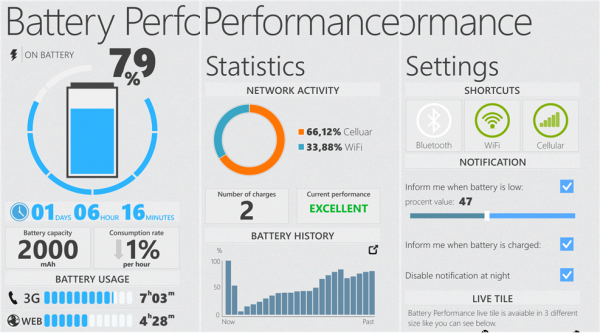
A battery is essentially a device that stores chemical energy that is converted into electricity. The Soviet Union decided to push forward with plans to develop these mobile devices. That may seem impressive, but that number accounted for only 9% of the total worldwide number of mobile users. The technology continues to make significant strides in the amount of time the device takes to charge a cell phone. Up to this point, scientists, inventors and battery companies are continually finding ways to discover how to make use of our available resources to store electricity. The signal was transmitted by radio waves. The inventor was working for the Eveready Battery Co.
Battery History

Today, you can find 6 cell or 9 cell laptop batteries that can run your laptop for hours on a single recharge. Samsung Galaxy Note — 2011 The Galaxy Note is on this list because Samsung had the foresight to build a stupidly large smartphone. His parents had lost two children to tuberculosis, and they insisted that the best way to save their last child was to leave England. These arcs were made of two different metals e. You can keep these batteries 100% charged all the time, which ensures that they are fully loaded when you are ready to go mobile.
The past, present and future of smartphone batteries
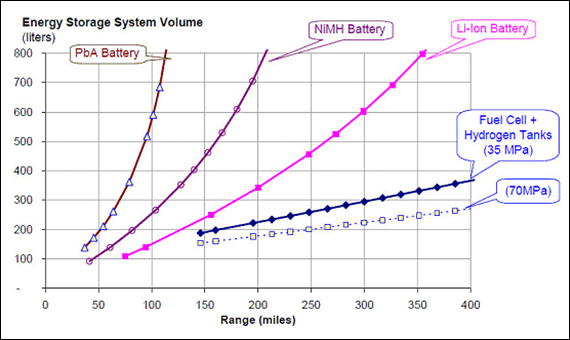
It became clear that 2G technology was not able to keep up. They were divided: some wanted to continue with the development of the battery, while others wanted to focus on the development of the cathode. Basically, batteries are small chemical reactors, with the reaction producing , ready to flow through the external device. The gravity cell consisted of a glass jar, in which a copper cathode sat on the bottom and a zinc anode was suspended beneath the rim. This came with advantages like better security and faster networking.
Charged up: the history and development of batteries

With this service, an estimated 5,000 customers placed approximately 30,000 calls each week. Thomas Edison's alkaline cell had iron as the anode material - and nickelic oxide as the cathode material +. Before long, demand had outstripped existing 2G technology. In these batteries, a lattice of graphite, stuffed with lithium ions, forms the anode. This had the transmitter and receiver in a simple handset. The introduction of nickel and lithium based batteries in the latter 20th century made the development of innumerable portable electronic devices feasible, from powerful to mobile phones. The telegraph had been a highly successful communication system for about 30 years before Bell began experimenting.
History of mobile phones

Read about the history of batteries and learn how they came to be the portable powerhouses they are today. This cell achieved very quick success in telegraphy, signalling and electric bell work. Batteries have been with us for a long time. Throughout the latter half of the 20th century, however, researchers would work on creating cellular networks that expanded cell service across America. Lithium-Ion Batteries are extremely popular in the technology industry for several reasons. The cell phones of today are also replacing our other gadgets, such as cameras and video cameras.