History of batteries facts. New York City History 2019-03-07
10 Interesting Battery Facts About Products You Use Every Day — BatteryPlex Blog

In comparison, very few Americans outside of cities had electricity at that time. In response to British aggressions, including the seizure of American trading ships and arming of Native American populations, the U. It was thought that it was used for metallic plating, rather than as a battery to generate electricity. In most other simpler chargers, the process typically takes overnight. The iconic Nokia 3310s battery capacity is 3 — 4 times less than an average smartphone today.
Battery History

Nevertheless, Edison's battery achieved great success in other applications such as electric and diesel-electric rail vehicles, providing backup power for railroad crossing signals, or to provide power for the lamps used in mines. He unwittingly started the world down the pathway of portable electricity. This battery type produced 1. There are a lot of batteries hitting the market today that provide individuals with ample benefits including a longer lifespan. Primary aluminum production 5% 7. Stephanie hails from East Tennessee, the global center for salamander diversity. That means you really have to get to know what these products can offer to you and what they are responsible for doing in your life.
10 Interesting Battery Facts About Products You Use Every Day — BatteryPlex Blog

These batteries hold their electrolyte in a solid polymer composite instead of in a liquid solvent, and the electrodes and separators are laminated to each other. Edison wanted to commercialise a more lightweight and durable substitute for the lead-acid battery that powered some early automobiles, and hoped that by doing so electric cars would become the standard, with his firm as its main battery vendor. The trend caused eye inflammation, tooth rot, baldness, and eventually, death. The cathode reacts with the electrolyte and wants electrons this is the positive or + end. Rechargeable batteries are recharged by reversing the chemical reaction that occurs within the battery.
History Of Batteries: A Timeline
Ayrton Practical Electricity; A Laboratory and Lecture Course for First Year. Apparently the recipe had some appeal: In the 1940s: 7 Up was the third best-selling soft drink in the world, according to Cadbury. The of 1812 was a high-voltage dry battery but capable of delivering only minute currents. Among these, Castle Clinton at The Battery, Castle Williams on Governors Island, and Fort Wadsworth on Staten Island still stand today. Alkaline batteries provided enough power to make way for portable radios, boom boxes and toys that beep and blink. It's used in the manufacture of aircraft and in certain batteries.
Battery Facts for Kids

The process is complex and there are very specific standards for efficiency and safety in place that makes battery creation time consuming and costly. It quickly became the battery of choice for the American and British telegraph networks, and was used until the 1950s. Around this same time, many big and small automakers began exploring options for alternative fuel vehicles, including electric cars. This is yet another reason why these products tend to cost much more than what many would assume to pay. Wires connected to either end of the stack produced a continuous stable current.
Battery Facts

Researchers witnessed the dying star ejecting lithium into space. But why was it ever there in the first place? It is also useful for things that moved around and cords would get in the way. What makes them so beneficial? There truly is a day for everything, which leaves us asking — when was the last time you checked the? Non-rechargeable batteries should not be charged as various harmful and substances can leak out, such as. A scientist named enhanced the lead-acid battery. Just a sample of these are: Reminder Lithium proved that drugs could successfully treat psychiatric disorders.
Battery Facts for Kids
These chemical reactions can be reversed by passing a reverse current through the battery, thereby recharging it. This becomes more essential as the batteries become larger, and frequently replacing them is not commercially viable. In the , sometimes called Grenet Cell due to the works of around 1859, the electrolyte was dilute sulphuric acid and the depolariser was chromic acid. Added to molten glass, it makes the glass lighter and stronger 4. Learn about the history of the hybrid -- from the to the.
10 Interesting Battery Facts About Products You Use Every Day — BatteryPlex Blog

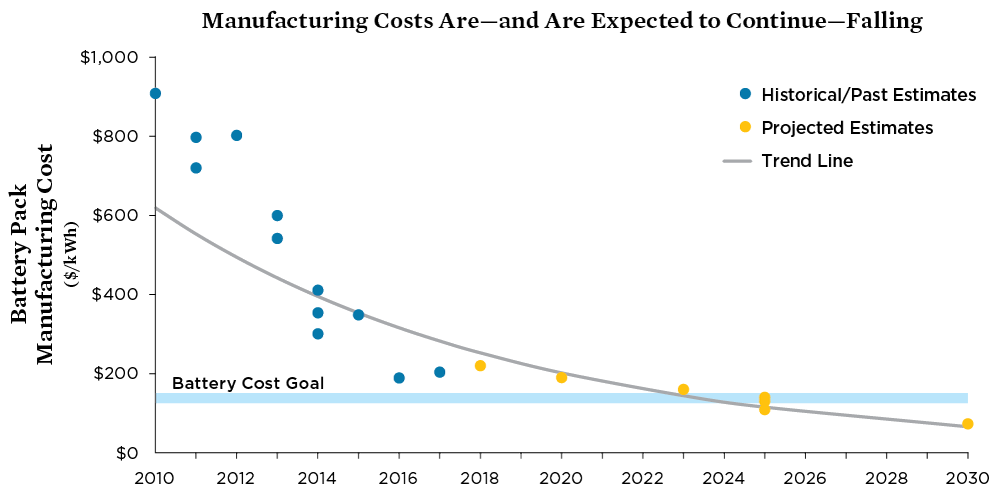
This in turn has helped lower the costs of electric vehicles, making them more affordable for consumers. One metal collects an excess of electrons, while the other metal loses electrons. Primary vs rechargeable batteries When the reaction that produces the flow of electrons cannot be reversed the battery is referred to as a primary battery. Written by: When it comes to , you probably do not think much about that little cylinder you are slipping into the back of the remote. The batteries can be mounted in any direction, even upside down. It is optimal for your lithium-ion smartphone battery to top it off and keep its charge between 40-80%, rather than let it drain 0% and charge it all the way up. The alkali metals provide one of the best examples of group trends in properties in the periodic table, with well characterized homologous behavior down the group.
Battery History

This battery was composed of a central zinc soaked into an earthenware vessel containing a solution of zinc sulfate. Lithium-ion and lithium-polymer batteries appear to have the greatest potential for growth. These are slim batteries and tend to offer a significant density, though they do not compare in terms of long life to some much heavier batteries. Automakers and other private businesses also installed their own chargers at key locations in the U. Aluminum coated with a conducting polymer that has a low index of refraction was used to connect to the cathode particle network. Motorola and Qualcomm may be interested in further investing 4.
The History of the Electric Car

Even Henry Ford, who was friends with Edison, partnered with Edison to explore options for a low-cost electric car in 1914,. The Daniell Cell was also the first battery to incorporate mercury, used to reduce. Is an effective power source for small, long-life batteries 5. The barrier was porous, which allowed to pass through but kept the solutions from mixing. But a rechargeable battery can only be recharged a given amount of time recharge life. A post-doc assigned by Chiang with the task of turning the insulator into a conductor discovered that by adding about 1% of a metal such as zirconium, titanium or niobium, the conductivity increased by eight orders of magnitude or 100 million times.